|
To know more about molecular weights, one must first become familiar with the concept of atomic weights. This method works quite well for many gases and volatile liquids, but it cannot be used for substances that decompose on heating, such as urea. The mass of the vapor is then calculated, and used in conjunction with the ideal gas law to determine the molecular weight of the liquid. 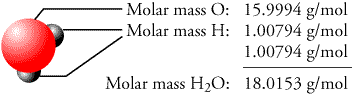
The flask is then allowed to cool, and the vapor condenses as air re-enters the flask. As the liquid evaporates, its vapor replaces the air in the flask. The flask is then heated to a temperature above the boiling point of the liquid. Experimentally, a few milliliters of a volatile liquid are placed in a stoppered flask containing a small orifice. The classical method of determining the molecular weight of a gas is to use density data. Unfortunately, Avogadro ’s ideas had little influence on the work of his contemporaries, and it was not until 1860 that another Italian scientist, Stanislao Cannizzaro (1826 –1910), pointed out that Avogadro ’s hypothesis could be used as a basis for determining atomic and molecular weights. In 1811, the Italian physicist Lorenzo Romano Amadeo Carlo Avogadro di Quaregua e di Cerreto (1776 –1856), known to posterity as Avogadro, concluded that equal volumes of all gases at the same temperature and pressure contain the same number of molecules. Although the law of Dulong and Petit proved a fair approximation for many elements, it was far from exact for many others, and it was not at all helpful in determining the atomic weights of gaseous elements. They suggested that the amount of heat required to raise the temperature of an atom of a solid material by a given amount should be independent of the type of atom, with the result that the gram atomic weight should be inversely proportional to the material ’s specific heat. 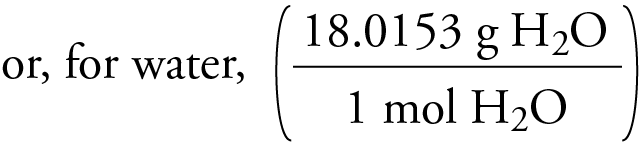
The first direct approach to determining molecular weight was proposed by two French scientists in 1819, Pierre Louis Dulong (1785 –1838) and Alexis-The ´r èse Petit (1791 –1820). 
In general, molecular weight can be determined by either chemical methods or mass spectrometry. As an example, the molecular weight of water, H 2O, is calculated as (2 × 1.00797) + (1 × 15.9994) = 18.0153, where 1.00797 and 15.9994 are the atomic weights of hydrogen (H) and oxygen (O) atoms, respectively. (Although molecular weight is often used, the more accurate terminology is molecular mass.) A molecule can be viewed as an entity of one or more different atoms bound together by some kinds of mutual interactions. Molecular weight (MW) is the sum of the atomic weights of the atoms in a molecule.
0 Comments
Leave a Reply. |
 RSS Feed
RSS Feed
